Magnetic Nanostructures
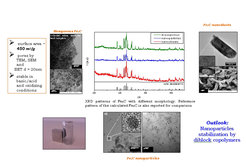
Magnetic nanomaterials possess special interest for a variety of applications, e.g. as magnetic recording material, sensors and in new nanoelectronic devices. Iron carbide, due to its higher stability against oxidation (compared to elemental iron), superior magnetic properties (compared to FeOx) and the extreme hardness is considered the ideal candidate to replace Fe° and FeOx. Crystalline Fe3C can be easily obtained by the soft urea pathway described above, using FeCl3 as metal precursors and heating treatment of the gel starting material at 700°C under N2 flow. Simply by playing with external parameters such as iron precursors, C-source, use of hard template, etc Fe3C with different morphology can be obtained.

Images of Fe3O4 and Fe3C particles (TEM and dispersions with PIL in water; left), dependence of the composition on the amount of urea (XRD; middle) and SQUID measurements of Fe3O4 and Fe3C (right).